Bonds That Form Between Water Molecules Are Called
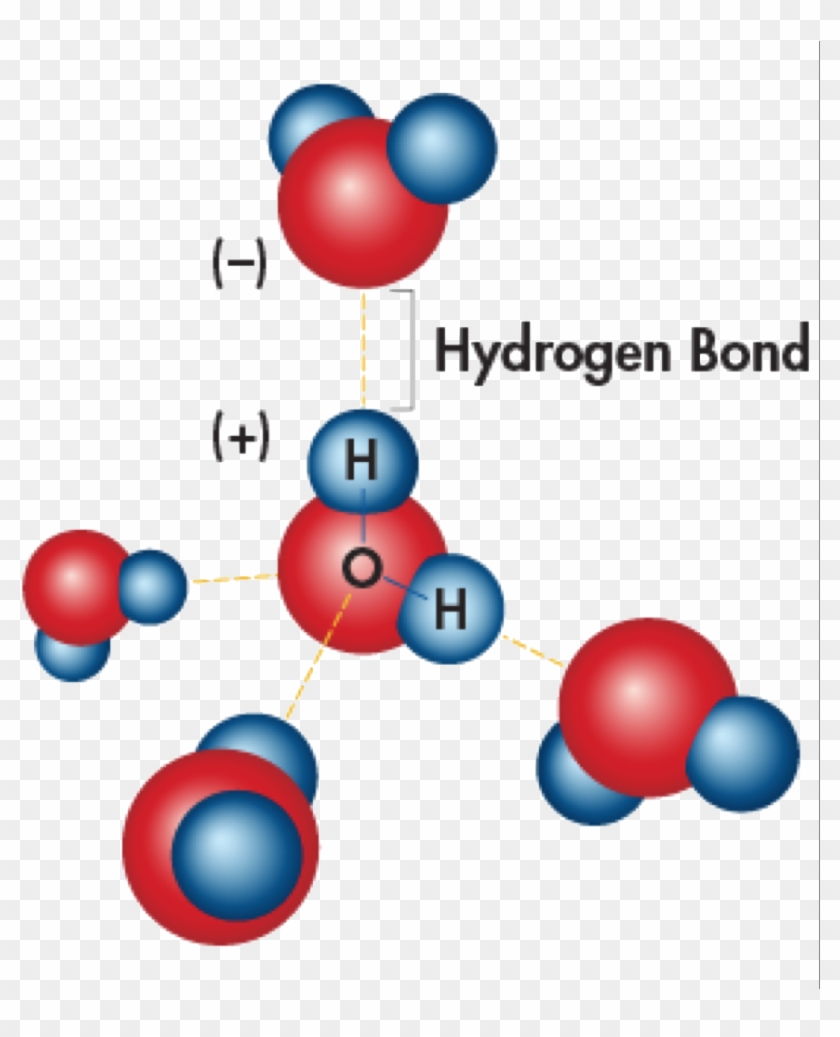
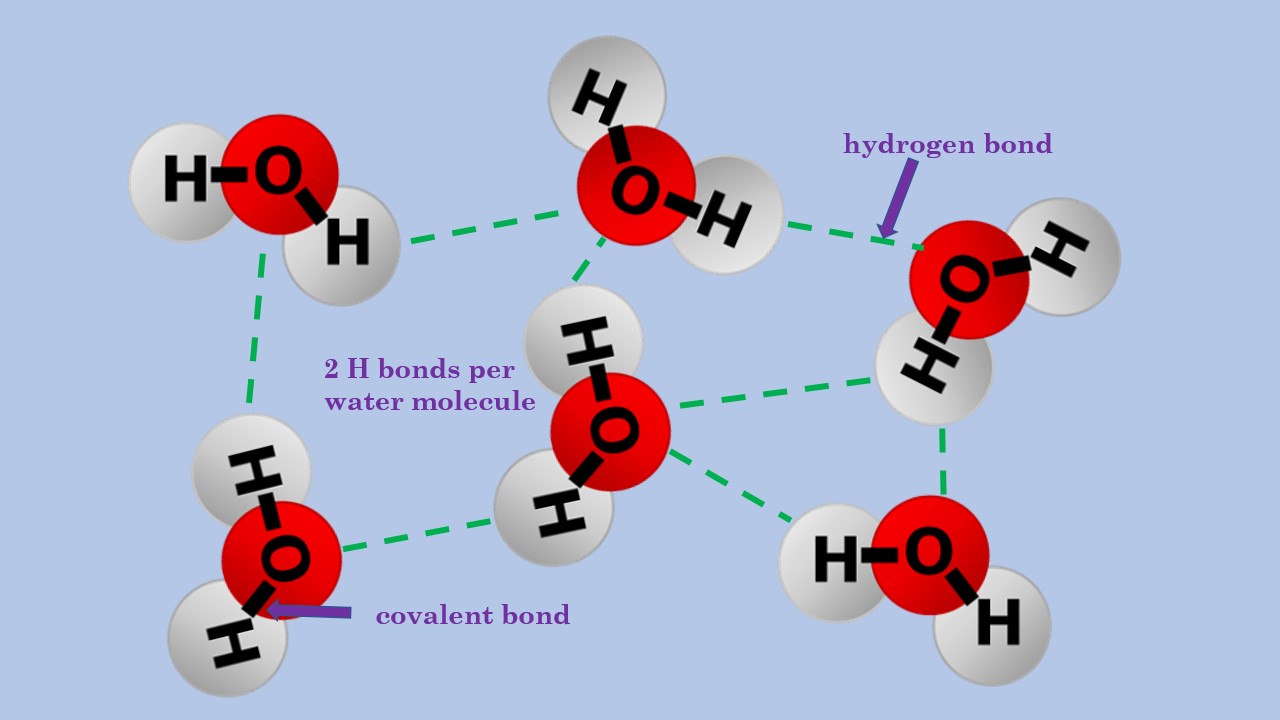
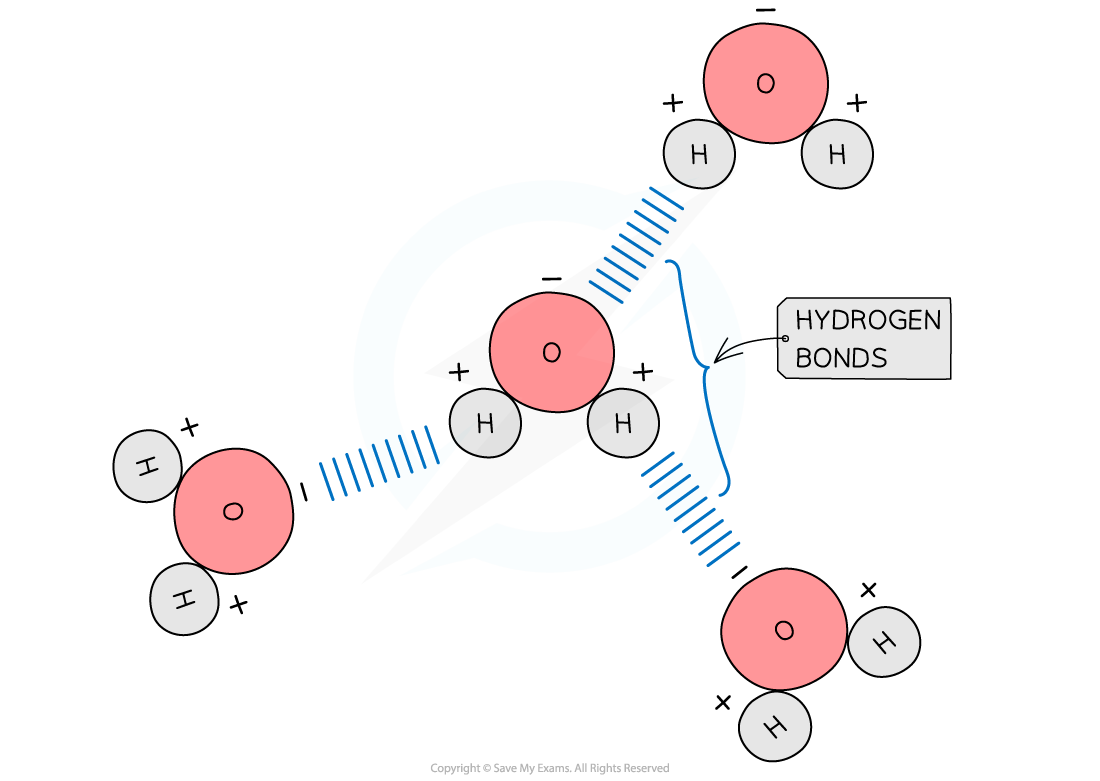
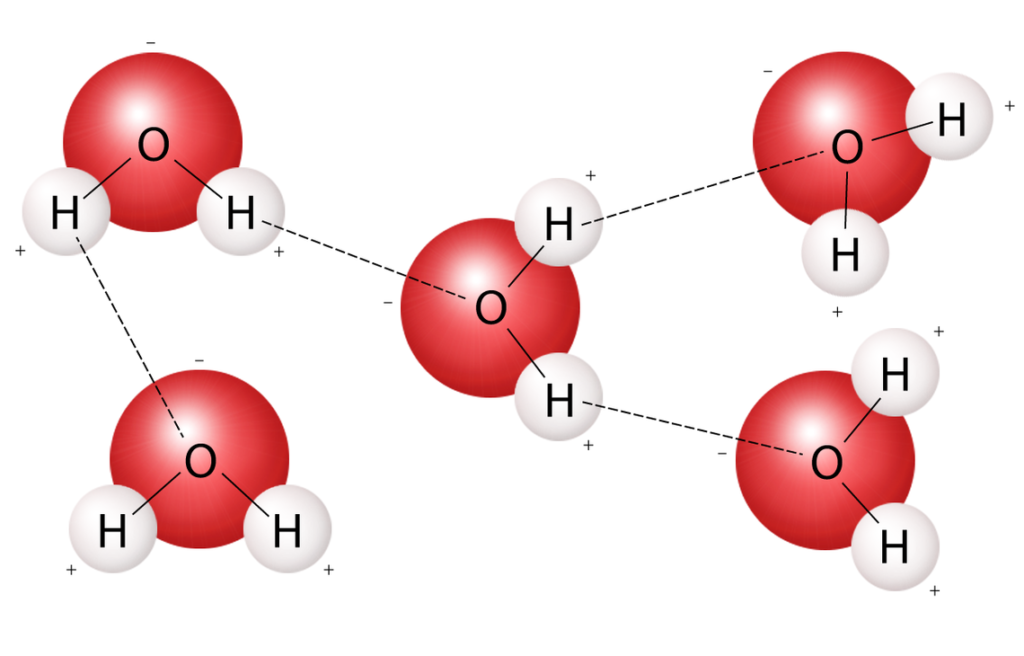
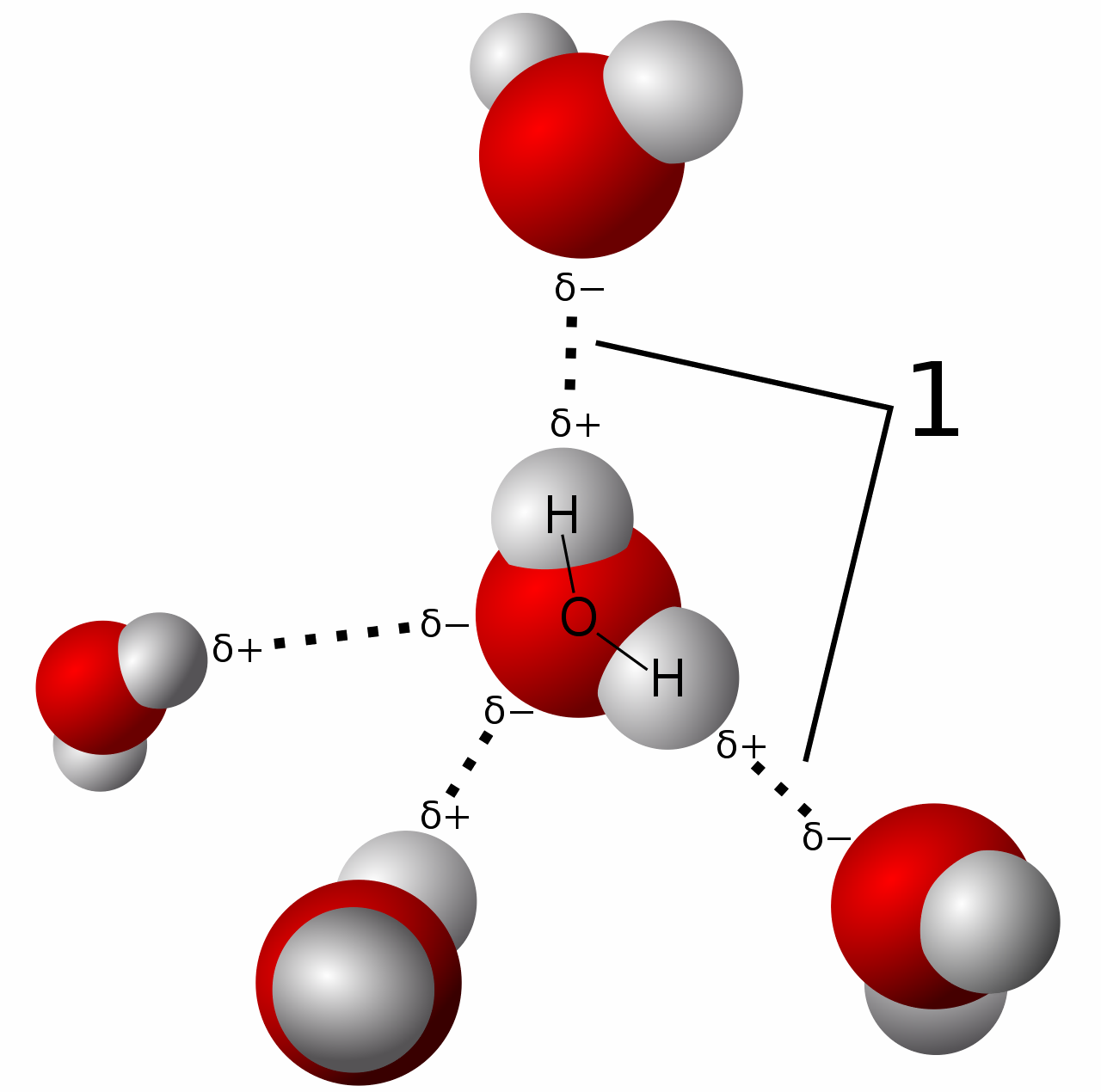
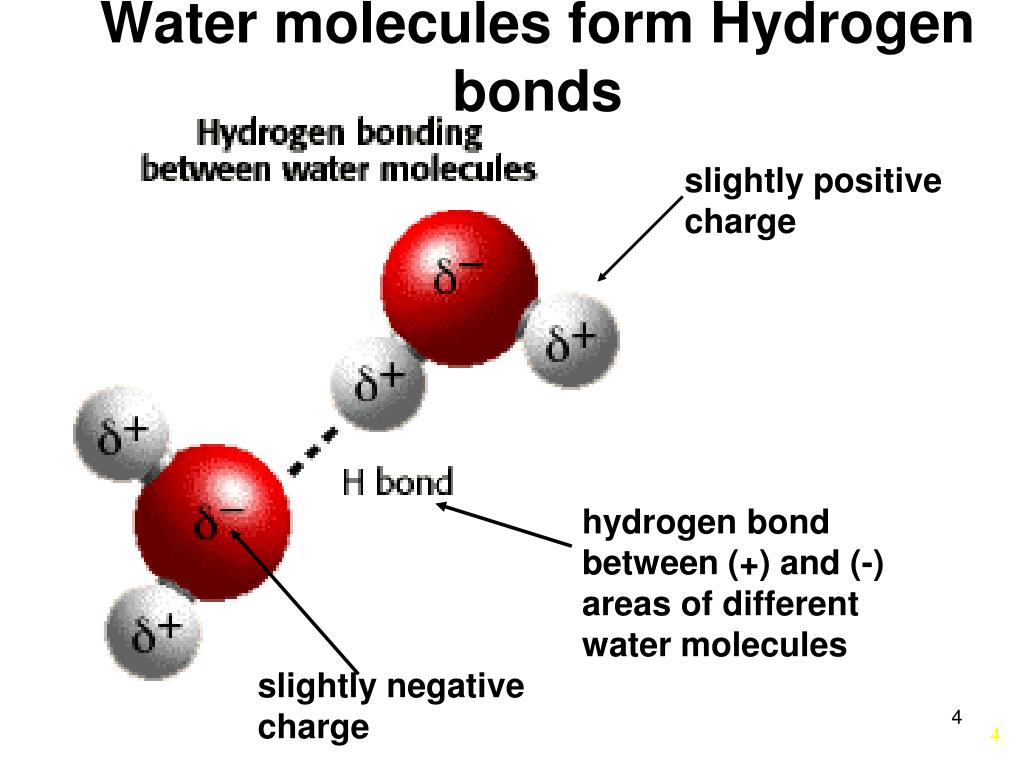
Bonds That Form Between Water Molecules Are Called - But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. Which of the following properties of.
Which of the following properties of. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding.
Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. Which of the following properties of.
Hydrogen Bonding Is The Effect Of Water Molecules Attracted
Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. Which of the following properties of. But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding..
The diagram shows hydrogen bonds between water molecules. Label the
The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. Which of the following properties of. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end.
How To Draw Hydrogen Bonding
But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. The hydrogen bonds that form between water.
IB DP Biology SL复习笔记2.1.3 Hydrogen Bonds翰林国际教育
But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. Which of the following properties of..
learning through art water molecules and hydrogen bonding
Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. Which of the following properties of. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water..
110 Properties of Water Lab Makeup) Professor St. John's
Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. Which of the following properties of. The.
Dipoles Of Water Molecules ALevel Biology Revision Notes
But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. Which of the following properties of. A drop of.
PPT Water Chemistry & Properties of Water PowerPoint Presentation
In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. But.
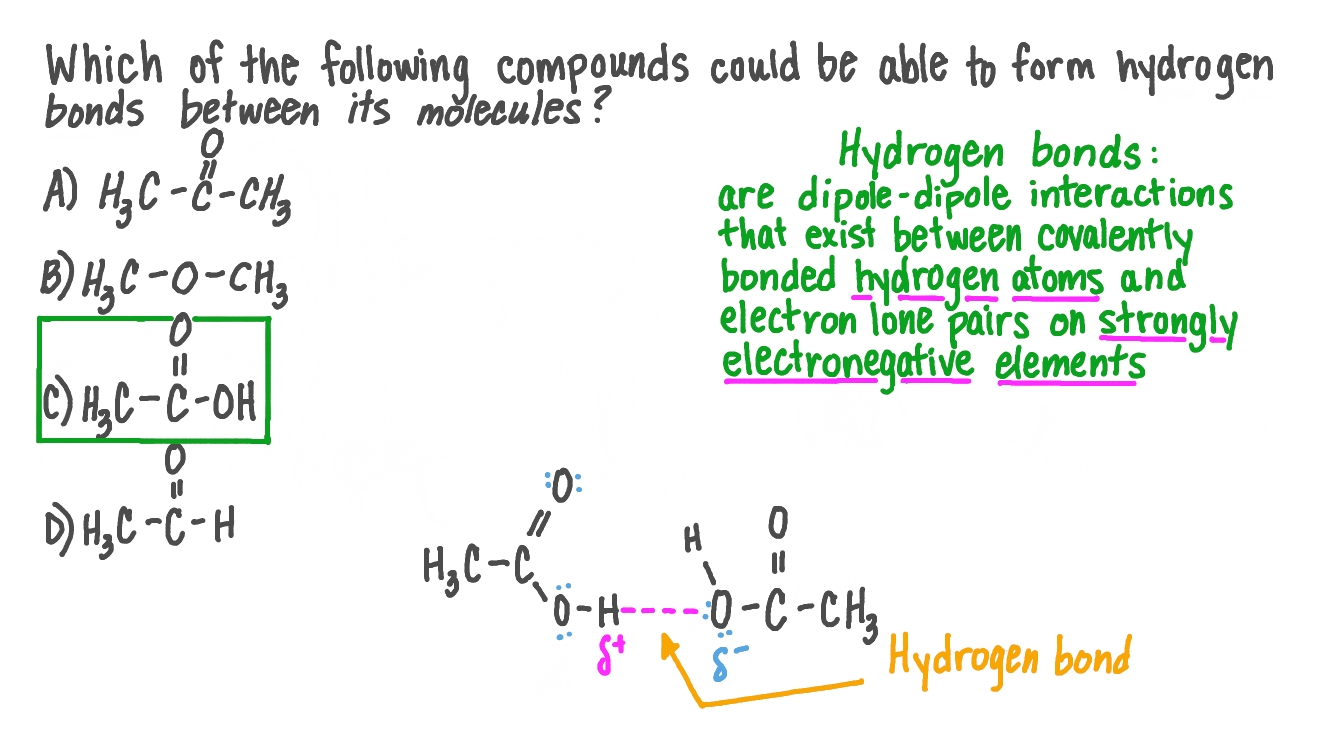
Question Video Identifying Which Compound Could Form Hydrogen Bonds
Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. Which of the following properties of. But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. In.
Biomolecules flashcards 1 How do hydrogen bonds form between water
A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. Which of the following properties of. Hydrogen bonds form between adjacent water molecules because the positively charged hydrogen end of one water molecule attracts. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent.
Which Of The Following Properties Of.
A drop of falling water is a group of water molecules held together by the hydrogen bonds between the molecules. But the intermolecular bonds, the bonds between water molecules, are the result of hydrogen bonding. The hydrogen bonds that form between water molecules account for some of the essential — and unique — properties of water. In the case of water, hydrogen bonds form between neighboring hydrogen and oxygen atoms of adjacent water molecules.