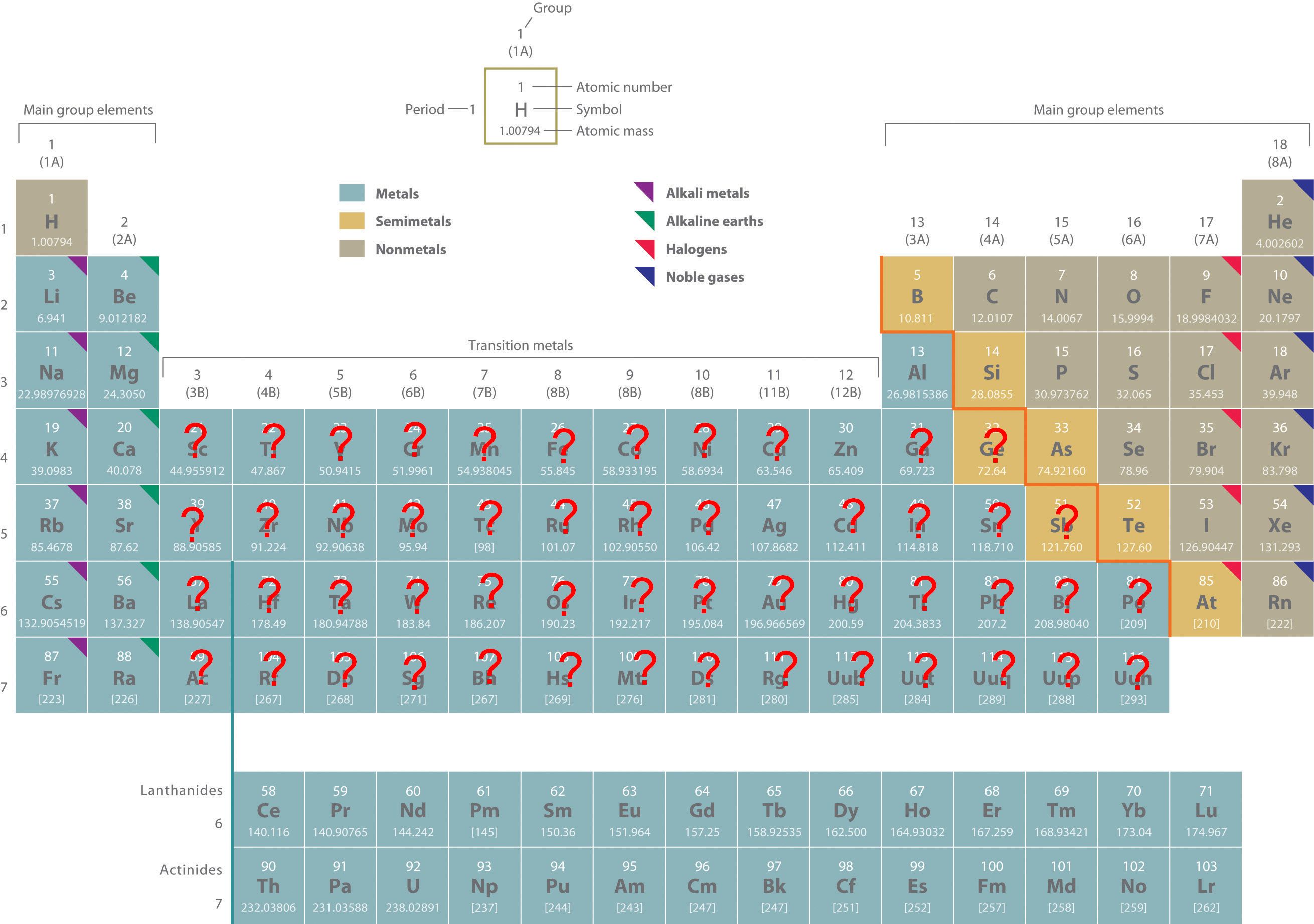
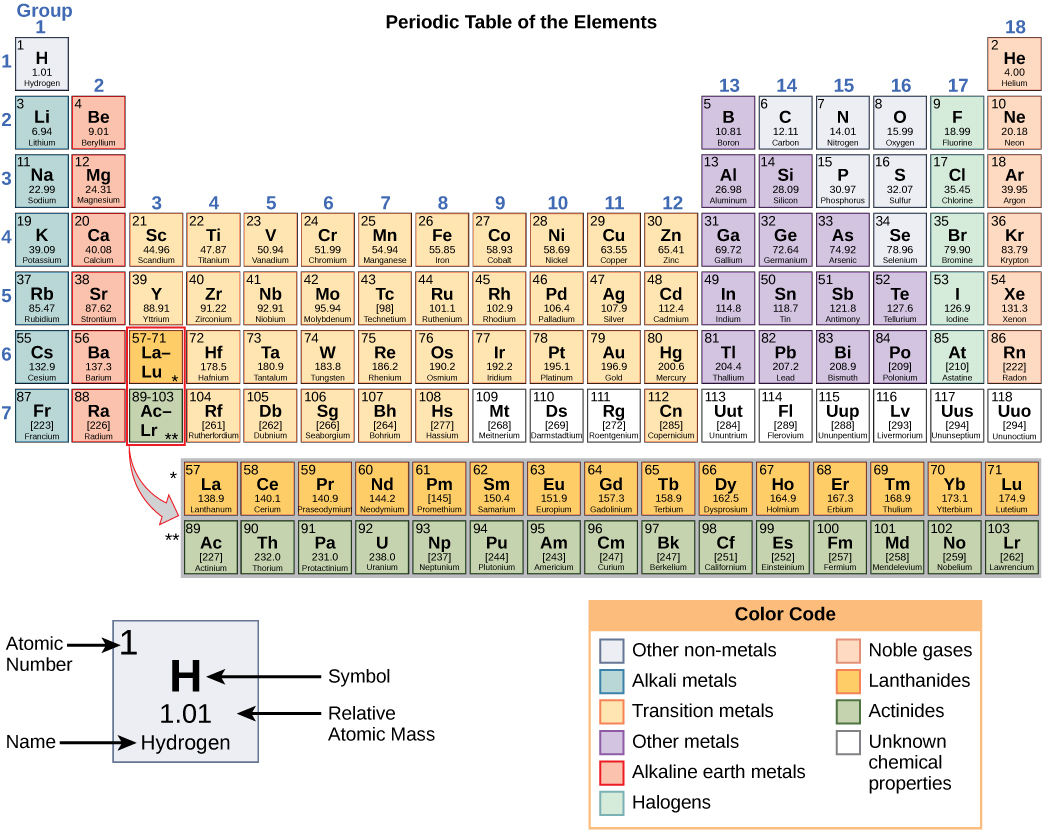
Do All B Group Elements Form More Than One Charge
Do All B Group Elements Form More Than One Charge - Study with quizlet and memorize. All group 2 elements (alkaline earth metals) lose two. All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All b group elements form more than one charge. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide?
All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? Study with quizlet and memorize. All b group elements form more than one charge.
All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All b group elements form more than one charge. Study with quizlet and memorize.
How Do Chemists Organize Information About Elements DeangelohasAtkinson
All b group elements form more than one charge. Study with quizlet and memorize. All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b.
Main Group Elements Definition
Study with quizlet and memorize. All group 2 elements (alkaline earth metals) lose two. All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All b group elements form more than one charge.
Element Charges Chart How to Know the Charge of an Atom
All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. Study with quizlet and memorize. All b group elements form more than one charge. All group 2 elements (alkaline earth metals) lose two.
metals tend to form what kind of ions Lombardi Bothe1936
Study with quizlet and memorize. All b group elements form more than one charge. All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b.
Periodic Table With Ionic Charges Labeled Elcho Table
All b group elements form more than one charge. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? Study with quizlet and memorize. All group 2 elements (alkaline earth metals) lose two. All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b.
Zinc group element chemistry Britannica
Study with quizlet and memorize. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge.
Properties of Elements Biology for NonMajors I
All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? Study with quizlet and memorize. All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge.
What Are The Transition Metals On The Periodic Table
Study with quizlet and memorize. All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All b group elements form more than one charge.
Naming Ionic Compounds
All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All group 2 elements (alkaline earth metals) lose two. All b group elements form more than one charge. Study with quizlet and memorize. All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b.
SOLVED Examining your labeled periodic table, which of the following
All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? Study with quizlet and memorize. All b group elements form more than one charge. All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All group 2 elements (alkaline earth metals) lose two.
All B Group Elements Form More Than One Charge.
All b group elements form more than one charge, what is the correct formula for vanadium(v) sulfide? All group 1 elements (alkali metals) lose one electron to form an ion with a 1+ charge b. All group 2 elements (alkaline earth metals) lose two. Study with quizlet and memorize.
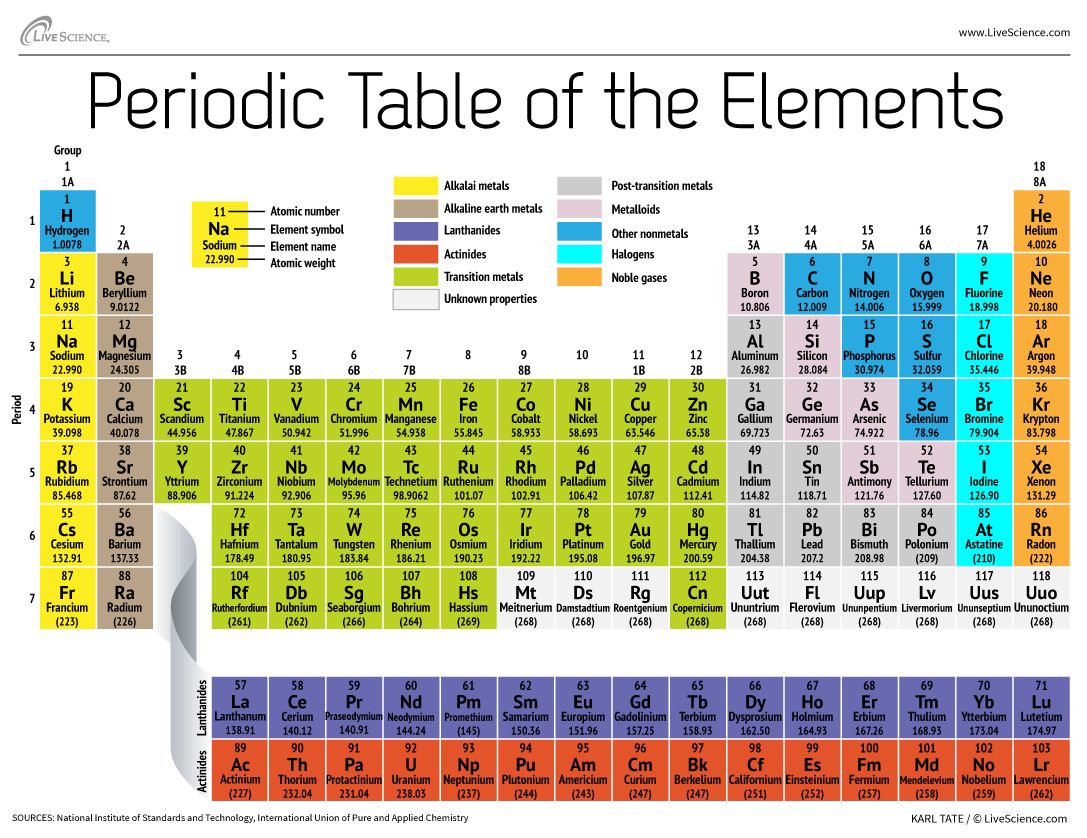
/the-periodic-table--digital-illustration--73016803-598b218ec41244001024af78.jpg)