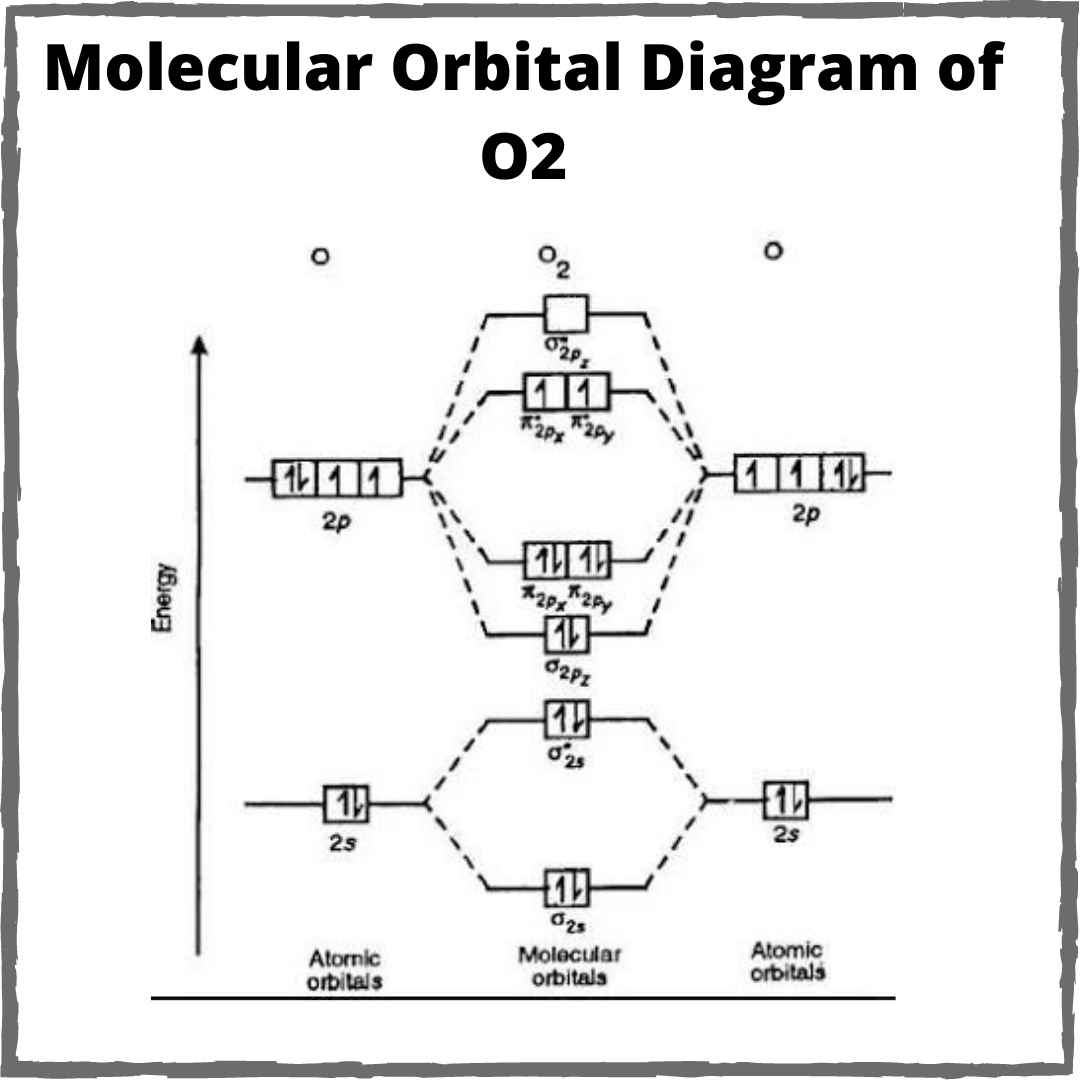
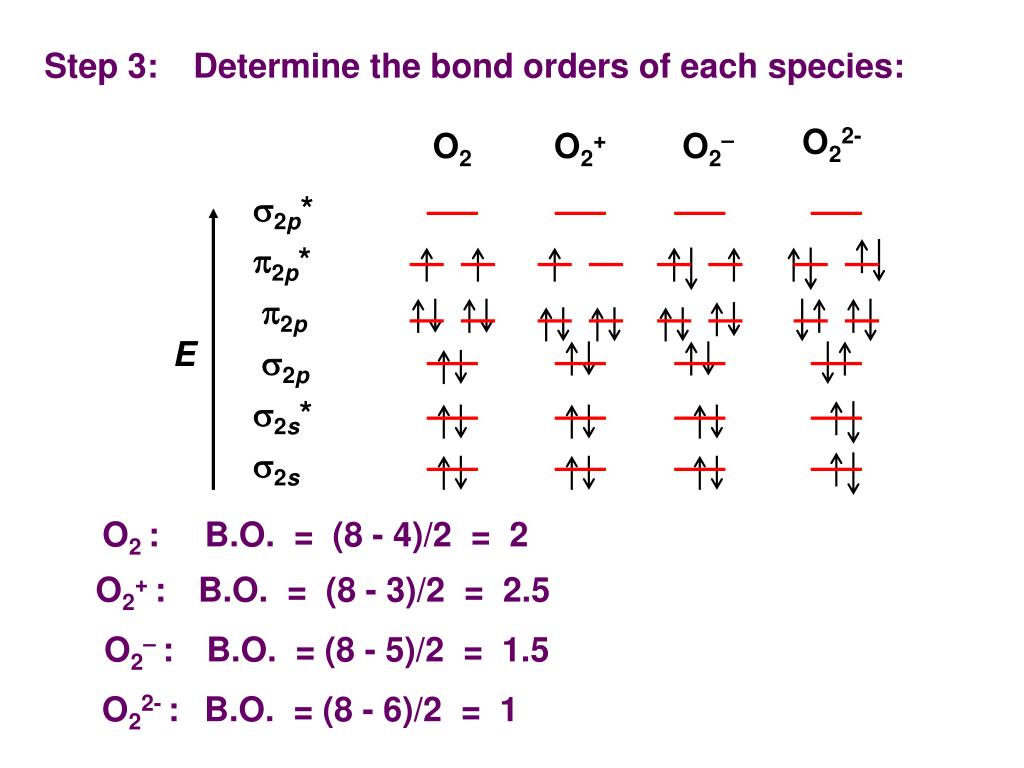
What Is The Bond Order For O2
What Is The Bond Order For O2 - Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For example, in diatomic nitrogen, n≡n, the bond.
For example, in diatomic nitrogen, n≡n, the bond. Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond.
Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For example, in diatomic nitrogen, n≡n, the bond.
38 o2 2 molecular orbital diagram Wiring Diagram Info
Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For example, in diatomic nitrogen, n≡n, the bond.
SOLVED Compare the stability of o2+ and o2 on the bases of molecular
For example, in diatomic nitrogen, n≡n, the bond. Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond.
How To Calculate Bond Order
Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For example, in diatomic nitrogen, n≡n, the bond.
Bond order for o2 Crack chemistry Crack Chemistry
Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For example, in diatomic nitrogen, n≡n, the bond.
Calculate the bond order in O2,O2^ ,O2^2 and O2^ + molecule.
For example, in diatomic nitrogen, n≡n, the bond. Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond.
O2 Molecular Orbital Diagram
For example, in diatomic nitrogen, n≡n, the bond. Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond.
BOND ORDER of O2 Bond order of SUPEROXIDE ION YouTube
Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For example, in diatomic nitrogen, n≡n, the bond.
What is meant by term bond order? Calculate the bond order of O2,o2
Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For example, in diatomic nitrogen, n≡n, the bond.
What is the bond order of the CO bonds in the carbonate ion? ECHEMI
For example, in diatomic nitrogen, n≡n, the bond. Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond.
Bond Order Is The Number Of Chemical Bonds Between A Pair Of Atoms And Indicates The Stability Of A Bond.
For example, in diatomic nitrogen, n≡n, the bond.