What Is The Product Of Hcl Naoh
What Is The Product Of Hcl Naoh - The type of salt produced depends on the. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The product of neutralization is a salt and water.
The type of salt produced depends on the. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The product of neutralization is a salt and water.
The product of neutralization is a salt and water. The type of salt produced depends on the. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible.
0 1 N Naoh Preparation How To Prepare And Standardize 0 1 N Sodium
The product of neutralization is a salt and water. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The type of salt produced depends on the.
20ML of 1N HCl reacted with 30ml of 0.5N Naoh, the normality of the
A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The product of neutralization is a salt and water. The type of salt produced depends on the.
HOW TO ACCEPT HCL OFFER LETTER hcl offer letter hcl onboarding
The type of salt produced depends on the. The product of neutralization is a salt and water. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible.
Acid/Base Neutralization Reaction for NaOH + HCl (Sodium hydroxide
The type of salt produced depends on the. The product of neutralization is a salt and water. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible.
Enthalpy of neutralization for both HNO3 and HCl with NaOH is 57.1kJ
The product of neutralization is a salt and water. The type of salt produced depends on the. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible.
TJD HCL TJD Shop
A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The product of neutralization is a salt and water. The type of salt produced depends on the.
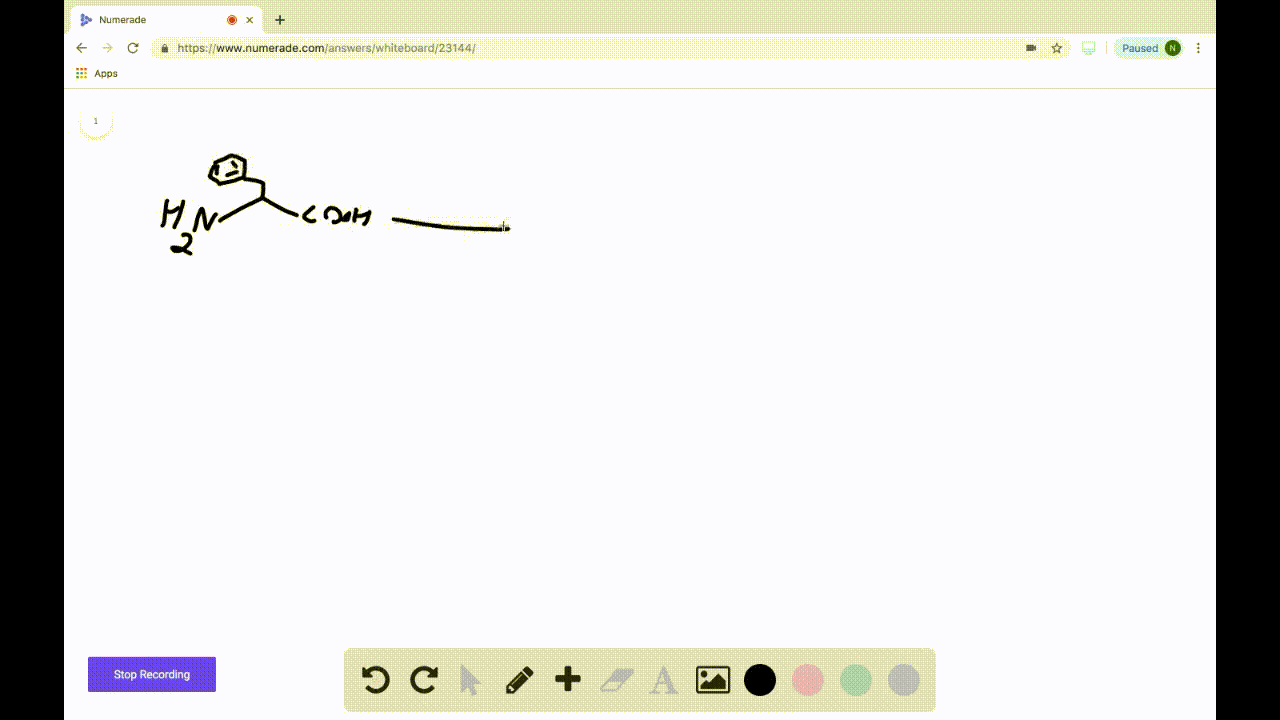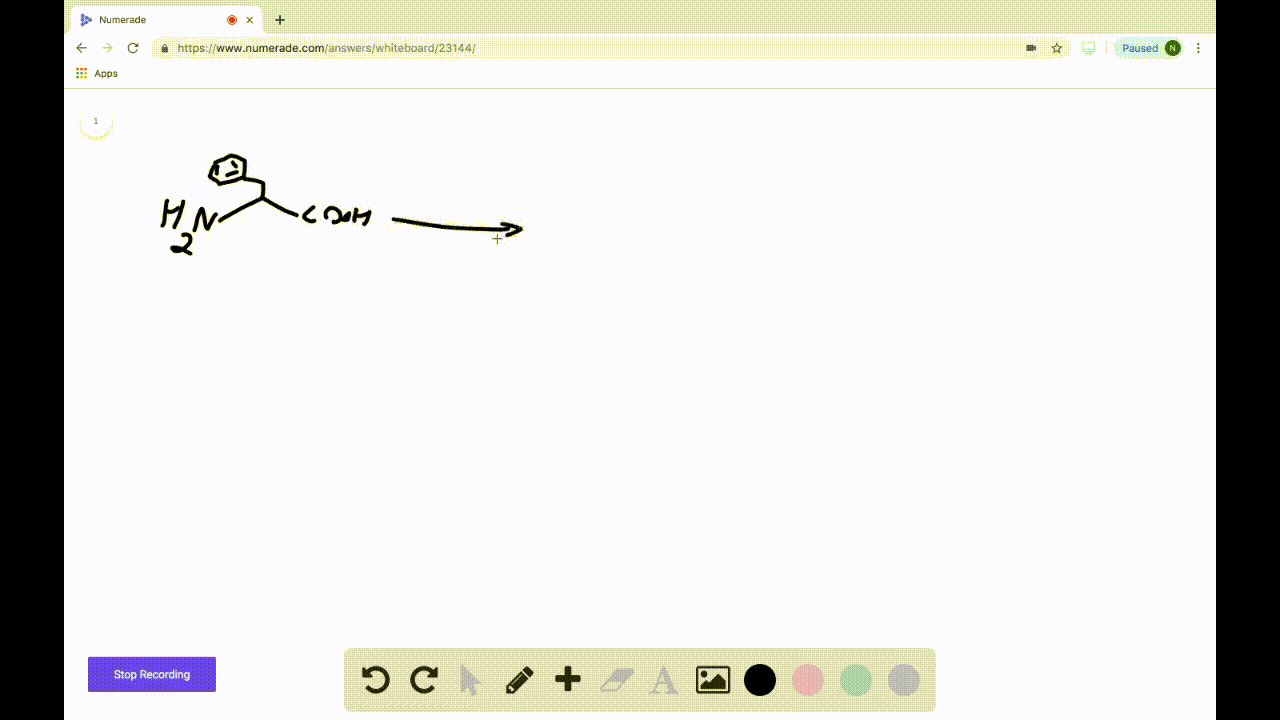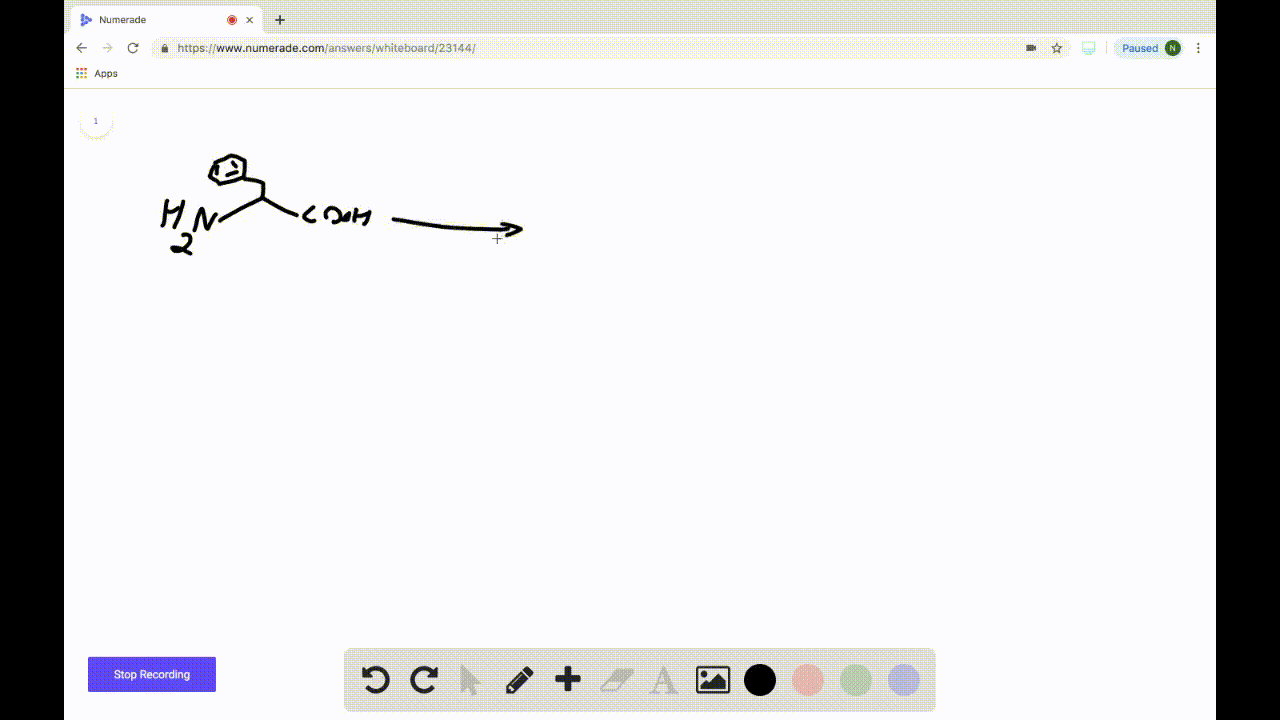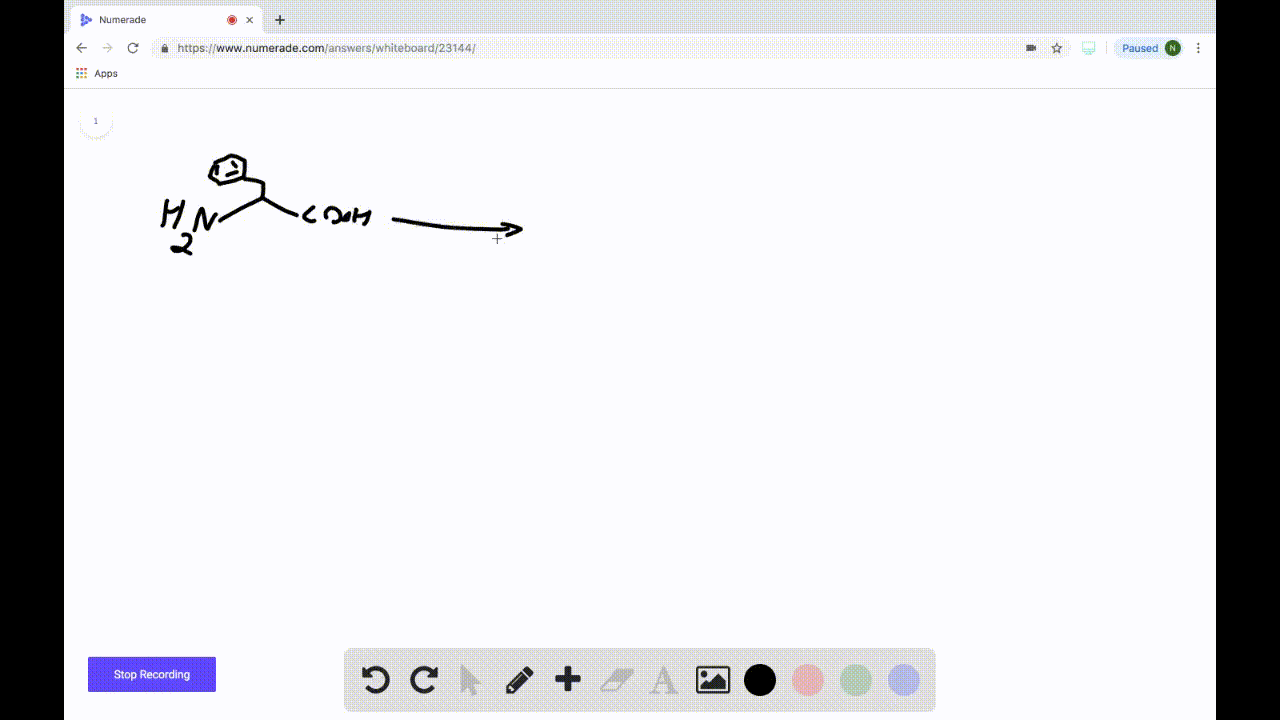
SOLVEDDraw the product formed when the following amino acid is treated
The type of salt produced depends on the. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The product of neutralization is a salt and water.
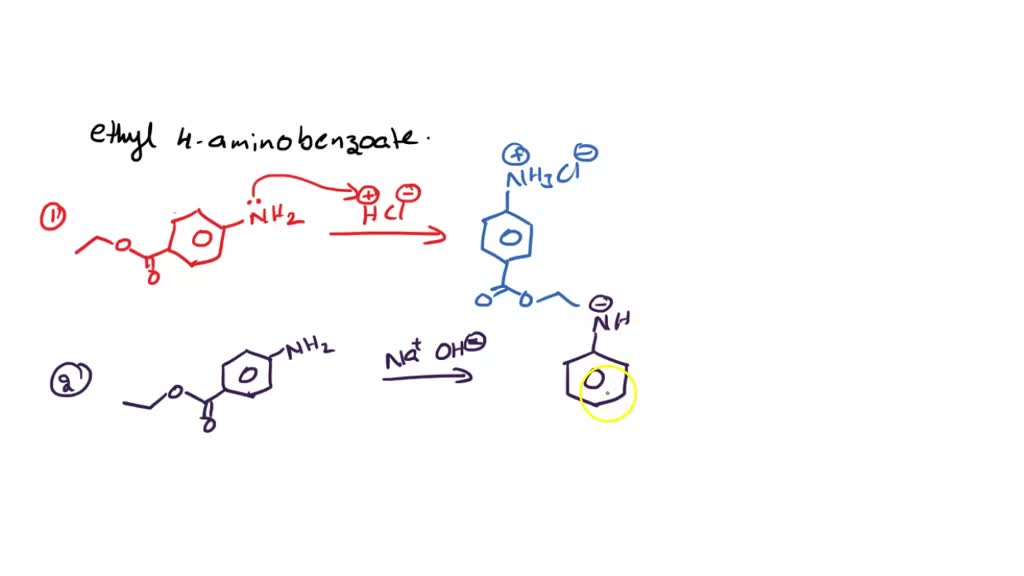
What are the reactions for ethyl 4aminobenzoate + HCl > ethyl 4
A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The product of neutralization is a salt and water. The type of salt produced depends on the.
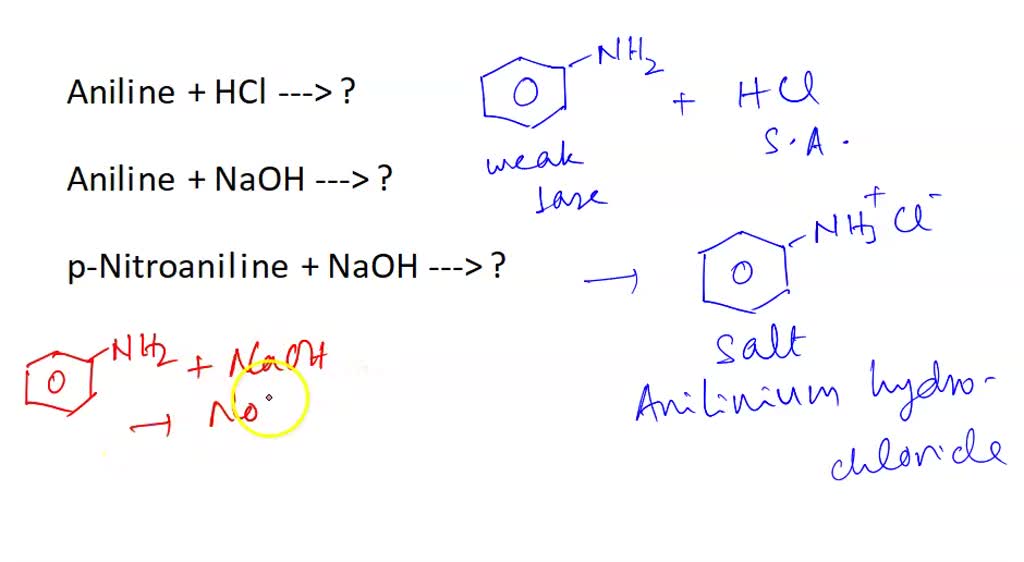
SOLVED Aniline + HCl —> ? Aniline + NaOH —> ? pNitroaniline + NaOH —> ?
The type of salt produced depends on the. The product of neutralization is a salt and water. A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible.
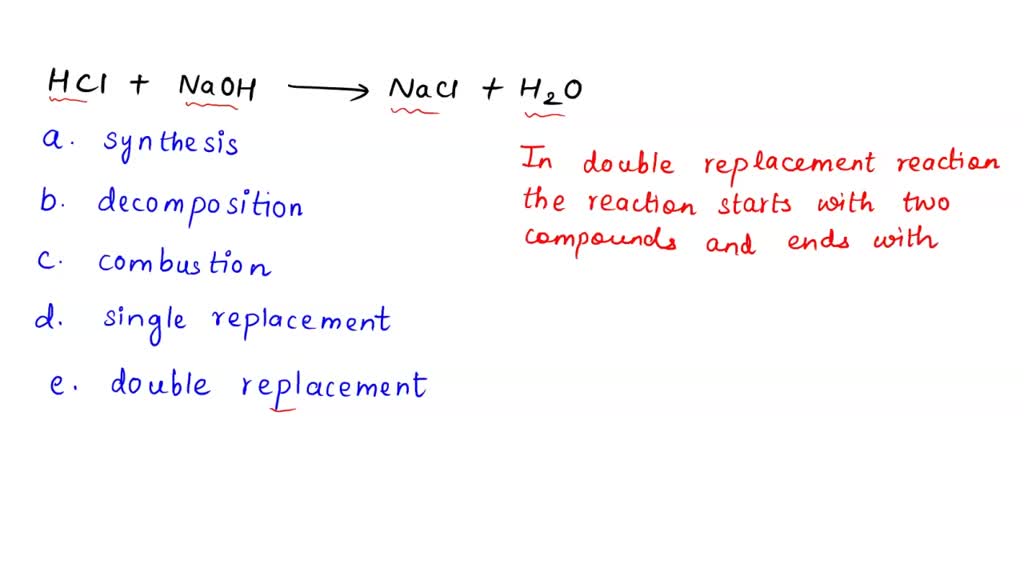
SOLVED HCl + NaOH —> NaCl + H2O What is the main classification of
A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The product of neutralization is a salt and water. The type of salt produced depends on the.
The Product Of Neutralization Is A Salt And Water.
A strong acid and a strong base, such as hcl(aq) and naoh(aq) will react to form a neutral solution since the conjugate partners produced are of negligible. The type of salt produced depends on the.